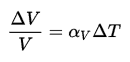Density of Heavy Water
Pure heavy water (D2O) has a density about 11% greater than water, but is otherwise physically and chemically similar.
This difference is caused by the fact, the deuterium nucleus is twice as heavy as hydrogen nucleus. Since about 89% of the molecular weight of water comes from the single oxygen atom rather than the two hydrogen atoms, the weight of a heavy water molecule, is not substantially different from that of a normal water molecule. The molar mass of water is M(H2O) = 18.02 and the molar mass of heavy water is M(D2O) = 20.03 (each deuterium nucleus contains one neutron in contrast to hydrogen nucleus), therefore heavy water (D2O) has a density about 11% greater (20.03/18.03 = 1.112).
Pure heavy water (D2O) has its highest density 1110 kg/m3 at temperature 3.98oC (39.2oF). Also heavy water differs from most liquids in that it becomes less dense as it freezes. It has a maximum of density at 3.98 °C (1110 kg/m3), whereas the density of its solid form ice is 1017 kg/m3. It must be noted, the change in density is not linear with temperature, because the volumetric thermal expansion coefficient for water is not constant over the temperature range.
We hope, this article, Density of Heavy Water, helps you. If so, give us a like in the sidebar. Main purpose of this website is to help the public to learn some interesting and important information about thermal engineering.