Thermal engineering is a specialized discipline of mechanical engineering that deals with the movement of heat energy and transfer. Since the energy can be transformed between two mediums or transferred into other forms of energy, a thermal engineer must have knowledge of thermodynamics and the process to convert generated energy from thermal sources into chemical, mechanical, or electrical energy.
One or more of the following disciplines may be involved in solving a particular thermal engineering problem:
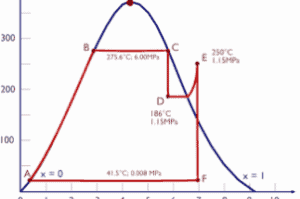
Thermodynamics
Fluid Mechanics
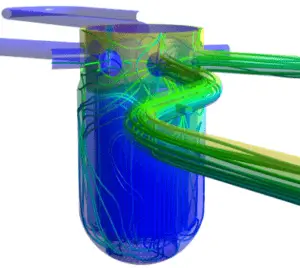
Fluid mechanics is the branch of thermal engineering concerned with the mechanics of fluids (liquids, gases, and plasmas) and the forces on them. It can be divided into fluid statics, the study of fluids at rest; and fluid dynamics. Fluid dynamics is a subdiscipline of fluid mechanics that deals with fluid flow. Fluid dynamics is one of the most important of all areas of physics. Life as we know it would not exist without fluids, and without the behavior that fluids exhibit. The air we breathe and the water we drink (and which makes up most of our body mass) are fluids. Fluid dynamics has a wide range of applications, including calculating forces and moments on aircraft (aerodynamics), determining the mass flow rate of water through pipelines (hydrodynamics).
Fluid dynamics is an important part of most industrial processes; especially those involving the
transfer of heat. In nuclear reactors the heat removal from the reactor core is accomplished by passing a liquid or gaseous coolant through the core and through other regions where heat is generated. The nature and operation of the coolant system is one of the most important considerations in the design of a nuclear reactor.
Heat Transfer and Mass Transfer
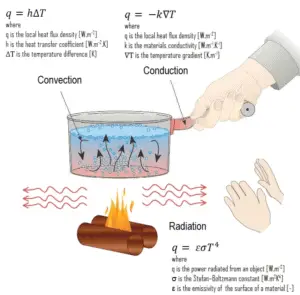
- Heat Conduction. Heat conduction, also called diffusion, occurs within a body or between two bodies in contact. It is the direct microscopic exchange of kinetic energy of particles through the boundary between two systems. When an object is at a different temperature from another body or its surroundings
- Heat Convection. Heat convection depends on motion of mass from one region of space to another. Heat convection occurs when bulk flow of a fluid (gas or liquid) carries heat along with the flow of matter in the fluid.
- Thermal Radiation. Radiation is heat transfer by electromagnetic radiation, such as sunshine, with no need for matter to be present in the space between bodies.
In engineering, the term convective heat transfer is used to describe the combined effects of conduction and fluid flow. At this point, we have to add a new mechanism, which is known as advection (the transport of a substance by bulk motion). From the thermodynamic point of view, heat flows into a fluid by diffusion to increase its energy, the fluid then transfers (advects) this increased internal energy (not heat) from one location to another, and this is then followed by a second thermal interaction which transfers heat to a second body or system, again by diffusion.