Prandtl Number of Water and Air
Prandtl Number of Water and Air
Air at room temperature has a Prandtl number of 0.71 and for water at 18°C it is around 7.56, which means that the thermal diffusivity is more dominant for air than for water. For a Prandtl number of unity, the momentum diffusivity equals the thermal diffusivity and the mechanism and rate of heat transfer are similar to those for momentum transfer. For many fluids, Pr lies in the range from 1 to 10. For gases, Pr is generally about 0.7.
Water and steam are a common fluid used for heat exchange in the primary circuit (from surface of fuel rods to the coolant flow) and in the secondary circuit. It used due to its availability and high heat capacity, both for cooling and heating. It is especially effective to transport heat through vaporization and condensation of water because of its very large latent heat of vaporization.
A disadvantage is that water moderated reactors have to use high pressure primary circuit in order to keep water in liquid state and in order to achieve sufficient thermodynamic efficiency.
Prandtl Number
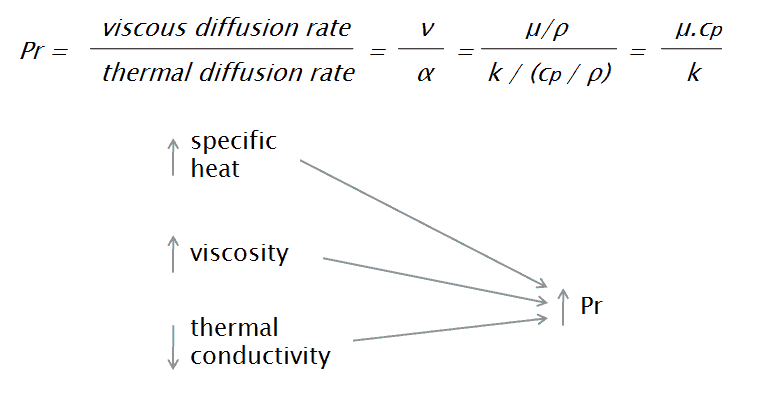
The Prandtl number is a dimensionless number, named after its inventor, a German engineer Ludwig Prandtl, who also identified the boundary layer. The Prandtl number is defined as the ratio of momentum diffusivity to thermal diffusivity. The momentum diffusivity, or as it is normally called, kinematic viscosity, tells us the material’s resistance to shear-flows (different layers of the flow travel with different velocities due to e.g. different speeds of adjacent walls) in relation to density. That is, the Prandtl number is given as:
where:
ν is momentum diffusivity (kinematic viscosity) [m2/s]
α is thermal diffusivity [m2/s]
μ is dynamic viscosity [N.s/m2]
k is thermal conductivity [W/m.K]
cp is specific heat [J/kg.K]
ρ is density [kg/m3]
Small values of the Prandtl number, Pr << 1, means the thermal diffusivity dominates. Whereas with large values, Pr >> 1, the momentum diffusivity dominates the behavior. For example, the typical value for liquid mercury, which is about 0.025, indicates that the heat conduction is more significant compared to convection, so thermal diffusivity is dominant. When Pr is small, it means that the heat diffuses quickly compared to the velocity.
In comparison to Reynolds number, the Prandtl number is not dependent on geometry of an object involved in the problem, but is dependent solely on the fluid and the fluid state. As such, the Prandtl number is often found in property tables alongside other properties such as viscosity and thermal conductivity.
We hope, this article, Prandtl Number of Water and Air, helps you. If so, give us a like in the sidebar. Main purpose of this website is to help the public to learn some interesting and important information about thermal engineering.